Fascination About Herbalife
Fascination About Herbalife
Blog Article
The Facts About Herbalife Revealed
Table of ContentsThe Buzz on HerbalifeHow Herbalife can Save You Time, Stress, and Money.Some Of HerbalifeThe Greatest Guide To Herbalife
Natural medicines are normally sold as food supplements, but a typical regulatory structure does not exist in different nations. Consequently, details on clinical indications for their use, efficiency, and safety and security are affected by the conventional experience available in each area. A brief summary of the regulation in USA, copyright, and Europe is offered in this section, and might be utilized to lead the lawful elements of the organic medicine industry in various other countries.Dietary supplements do not need approval from the Fda (FDA) prior to they are marketed (FDA 2010. herbalife products and pricing. Under DSHEA, organic medicines, which are classified as dietary supplements, are presumed secure, and the FDA does not have the authority to need them to be authorized for security and efficiency before they enter the market, which holds true for medications
Nevertheless, a dietary supplement producer or representative of a supplement with a "new nutritional component," that is, an ingredient that was not marketed in the USA before October 1994, may be called for to undergo premarket review for safety and security data and other information. All residential and foreign companies that manufacture package labels or hold dietary supplements need to comply with the FDA's present great manufacturing technique (GMP) policies, which detail procedures for guaranteeing the quality of supplements intended for sale (FDA 2010; Gao 2010).

Not known Facts About Herbalife
In order to be given a license, detailed details on the medical ingredients, resource, effectiveness, nonmedicinal active ingredients, and suggested use demands to be equipped. Once an item has been granted a license, it will certainly bear the certificate number and follow common labeling demands to guarantee that customers can make educated options.
In addition, GMPs need to be used to guarantee item safety and top quality. https://www.tripadvisor.com/Profile/isaacsQ4912AQ. This requires that suitable criteria and methods relating to the manufacture, storage space, managing, and distribution of all-natural health and wellness items be met. The GMPs are made to be end result based, guaranteeing risk-free and high-quality products, while offering the versatility to carry out top quality control systems proper to the product line and business
In Europe, the European Directive 2004/24/EC released in 2004 by the European Parliament and by the Council of Europe provides the guidelines for the use of organic medications (Calapai 2008 (herbalife protein powder). The instruction develops that herbal medicines released on the market demand authorization by the national regulatory authorities of each European nation and that these items have to have an identified level of safety and anonymous effectiveness (Calapai 2008
When it come to the production of these products and their top quality, products have to satisfy the same demands as applications for a marketing permission. Information is based on the accessibility of modern sciencebased public essays in the European Pharmacopeia and their matchings developed by the pharmaceutical industry. The standards advanced permit not only to specify the quality of items but likewise to remove hazardous compounds, debauchment, and contamination.
Some Ideas on Herbalife You Should Know
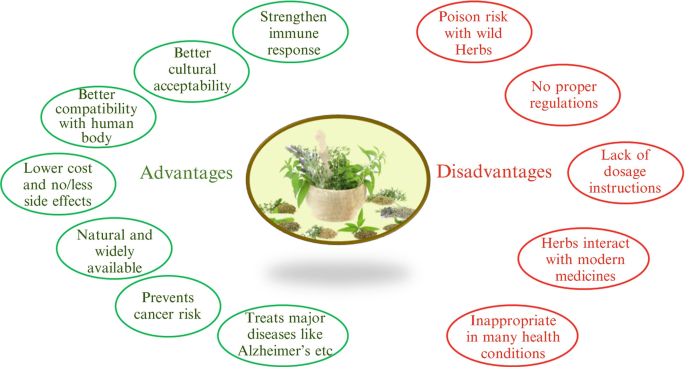
To separate each energetic component from each herb would be immensely lengthy at a high cost, making it not cost-efficient for makers (Richter 2003. Another issue is that despite the popularity of botanical dietary and herbal supplements, some organic items on the market are most likely to be of low high quality and suspicious effectiveness, also if the herb has been shown to have an impact in controlled researches making use of premium product
Herbs might well have unwanted side impacts, there are no set "doses," and herbdrug or herbherb interactions are feasible. A significant theoretical advantage of botanicals over standard single-component medications is the existence of multiple active substances that together can provide a potentiating impact that might not be possible by any type of single substance.

Excitement About Herbalife
The quality control of herbal medications has a direct impact on their safety and effectiveness (Ernst, Schmidt, and Larger 2005; Ribnicky et al. 2008. But, there is little data on the make-up and quality of the majority of herbal medicines not just because of absence of ample plans or federal government requirements but additionally as a result of an absence of sufficient or accepted study approach for reviewing standard medicines (THAT 2001; Kantor 2009. To isolate each energetic ingredient from each natural herb would be profoundly lengthy at a high cost, making it not cost-efficient for producers (Richter 2003. One more issue is that despite the popularity of agricultural dietary and herbal supplements, some natural products on the marketplace are most likely to be of poor quality and suspicious efficacy, even if the herb has been shown to have an impact in regulated researches making use of high-grade item
Herbs may well have unwanted side impacts, there are no set "doses," and herbdrug or herbherb communications are possible. A significant theoretical advantage of botanicals over standard single-component medicines is the presence of several active substances that together can give a potentiating impact that may not be possible by any kind of single substance.
Substances that are identified by activity-guided fractionation must be examined in appropriate pet models to verify in vivo activity. Ideally, the structure of the total botanical essence should be standardized and devoid of any kind of possible risks, and plants need to be grown particularly for the manufacturing of botanical removes under regulated conditions and stem from an identified and uniform genetic resource with a taxonomic record of the genus, species, and cultivar or various other additional identifiers.
Report this page